Share this @internewscast.com
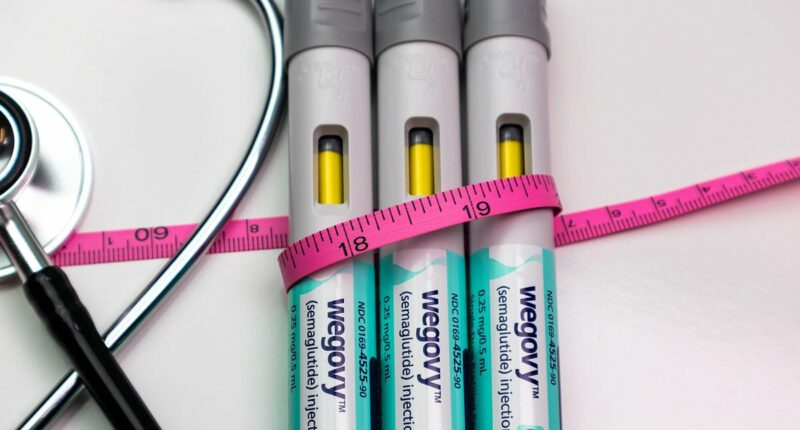
In an advancement for those using weight loss injections, a triple-dose version of the popular Wegovy jab has received official approval, allowing users to administer the maximum dose with just one injection. This development follows the decision by the Medicines and Healthcare products Regulatory Agency (MHRA) to approve a new Wegovy pen capable of delivering a 7.2mg dose of semaglutide in a single shot.
The introduction of this once-weekly mega dose eliminates the need for users to manage three separate 2.4mg injections, streamlining the process for those seeking to lose or maintain weight. Previously, the NHS prescribed Wegovy at a maximum dosage of 2.4mg, but with MHRA’s approval of the higher dosage earlier this year, the approach to weight management is set to become more efficient.
Manufacturers Novo Nordisk are expected to release these single-dose devices in the UK in about four to six weeks, thanks to the recent regulatory approval. The new device not only simplifies the dosing process but also includes an automatic mechanism that locks after use, ensuring a safer and more convenient experience for users.
The injection is already prescribed by the NHS at a maximum dose of 2.4mg, with the MHRA approving the higher dose of 7.2mg in January this year.
But the new ruling will see Novo Nordisk, the manufacturers of Wegovy, roll out single-dose devices in the UK within the next four-to-six weeks.
The new single-dose device contains an automatic dosing mechanism which locks after use, marking a practical change in how the highest maintenance dose can be taken – potentially making weight-loss more accessible.
Sebnem Avsar Tuna, general manager at Novo Nordisk said: ‘The approval of this new single-dose device provides an additional administration option for people in the UK who are prescribed the 7.2mg once weekly maintenance dose of Wegovy.
‘We welcome the the MHRA’s decision and will continue to work with healthcare professionals and partners to support its introduction in line with local prescribing arrangements.’

The maximum 7.2mg dose will be administered via one injection, rather than three smaller 2.4mg doses
The decision comes following the results of the STEP UP clinical trial, which found that adults treated with the stronger dose achieved average weight loss of up to 20.7 per cent when used alongside a healthy diet and regular exercise.
Around a third of participants – all of which had a BMI over 30, making them clinically obese – achieved weight loss of 25 per cent or more after 72 weeks.
Those given a placebo drug lost just 1.4 per cent of baseline bodyweight.
Common side-effects which resulted in discontinuation included gastrointestinal issues such as nausea, mainly occurring when the dose was escalated from 0.25mg – the standard starting dose – to 7.2mg.
Further analysis showed the higher 7.2 mg once-weekly dose could be used to achieve greater weight loss in people who have plateaued on the 2.4 mg maintenance dose for at least four weeks.
‘This approval represents another important step in Novo Nordisk supporting people living with obesity to achieve meaningful and significant weight loss,’ Mr Tuna added.
‘The addition of a new dose of semaglutide provides healthcare professionals with greater flexibility to tailor treatment for people living with obesity, supporting evidence-based approaches to improve health outcomes.’
However, other obesity experts have urged patients to exercise caution when it comes to increasing their dose.
He said: ‘Tripling the dose only gives a marginal extra benefit, but the dose increase is massive.
‘Going from 2.4mg to 7.2mg is a very big jump and I’m concerned many patients won’t tolerate such a high dose.’
He added: ‘Even if 7.2mg is approved, I suspect uptake will be low because of cost and side-effects – the top dose is already expensive.’
The new device will need to pass a NICE review to ensure cost effectiveness before it is rolled out on the NHS to eligible patients, but the new mega pen will be available on private prescription.
The MHRA added that patients should always use the medicine exactly as their prescriber tells them and to check with their doctor or prescriber if they are unsure.
As with any drug, the MHRA will continue to review the safety and effectiveness of Wegovy.
Anyone who suspects they are having side effects from the new stronger dose is encouraged to report it directly to the Yellow Card scheme.
Semaglutide – sold under Ozempic for diabetes and Wegovy for weight loss – has long been hailed as a monumental breakthrough on the war against obesity.
But experts have raised concerns about supply and demand, and suggested that the benefits may be short lived once treatments ends – warning that millions may need to stay on the jabs for life.