Share this @internewscast.com
On Wednesday, the FDA greenlit an innovative psoriasis treatment, promising potential relief for millions grappling with the skin disorder.
The regulatory body endorsed Johnson & Johnson’s Icotyde, an oral medication tailored for individuals aged 12 and up who are dealing with moderate to severe plaque psoriasis.
This groundbreaking pill offers a needle-free alternative to current therapies for the 8 million Americans affected by psoriasis, aiming to simplify treatment.
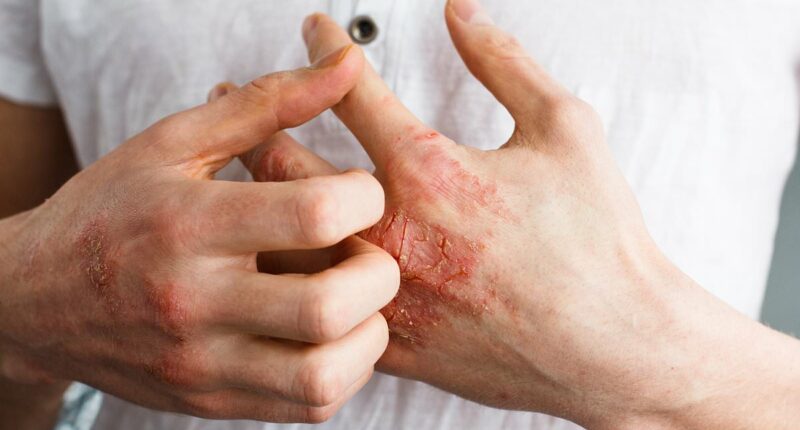
Psoriasis is an autoimmune disorder characterized by the rapid accumulation of skin cells, resulting in itchy, flaky, and inflamed patches known as plaques. It is triggered by a combination of genetic factors and environmental influences, such as stress or infections.
Though not life-threatening, psoriasis can lead to uncomfortable symptoms, such as itching, burning sensations, dry and cracked skin prone to bleeding, and swollen, painful joints.
Beyond physical discomfort, the condition often causes significant emotional challenges. Many sufferers face anxiety and embarrassment, which can exacerbate their mental health struggles.
Current treatment options include topical steroid creams, using artificial light to slow skin cell growth and injections, which can cost thousands each year even with insurance coverage and are less convenient than a daily pill.
Icotyde is the first and only oral treatment for psoriasis that targets the Interleukin-23 (IL‑23) receptor, a protein complex on immune cells that binds Il-23 cytokines, molecules that induce inflammation. While that inflammation helps fight infections, overactive levels lead to autoimmune diseases such as psoriasis.

The FDA has approved a new oral pill for psoriasis, an autoimmune condition that causes skin cells to build up rapidly, leading to itchy, flaky, scaly and inflamed skin patches, or plaques (stock image)
Dr John Reed, executive vice president, R&D, innovative medicine at J&J, said: ‘the approval of Icotyde represents a pivotal moment for people with plaque psoriasis.
‘At Johnson & Johnson, we are harnessing our scientific expertise to transform cutting-edge science into meaningful solutions for patients. Icotyde is a fundamentally different treatment with the potential to redefine what physicians and patients can expect from psoriasis treatment.’
The FDA’s approval follows positive results from four phase 3 clinical trials including more than 2,500 patients. The studies found that approximately 70 percent of patients achieved clear or almost clear skin after 16 weeks.
The research has shown Icotyde has a favorable safety profile with minor side effects, which include headache, nausea, cough, fungal infection and tiredness.
It’s still unclear if the drug is safe for pregnant or breastfeeding women.
J&J has not yet disclosed the list price for Icotyde, but injectable drugs that target IL-23 can run up to $100,000 per year. It’s also unclear if it will be covered by insurance.

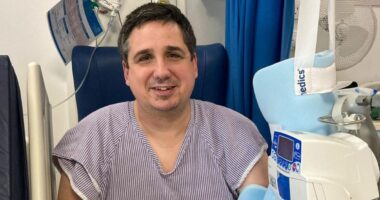
Icotyde pills, pictured above, were approved by the FDA for psoriasis
Dr Linda Stein Gold, director of dermatology clinical research at Henry Ford Health, said: ‘Icotyde delivers something unique in psoriasis treatment – combining skin clearance with a favorable safety profile in a once‑daily pill, making it an easy addition to a patient’s routine.
‘With new guidance from the International Psoriasis Council that clarifies when to move beyond cycling on topical treatments to systemic therapy, an innovative option like Icotyde is a potential game‑changer for many adult and adolescent patients.’
Psoriasis usually appears between ages 10 and 35, with peaks between ages 20 and 30 and 50 and 60. About one in three patients have a family member with the condition.
Flare-ups are often triggered by stress, skin injuries such as cuts or scrapes, infections and cold weather. Lifestyle factors that cause inflammation, such as smoking and heavy alcohol consumption, can also worsen symptoms.