Share this @internewscast.com
Need to know: Urgent recall of Ramipril blood pressure medication due to packaging mix-up
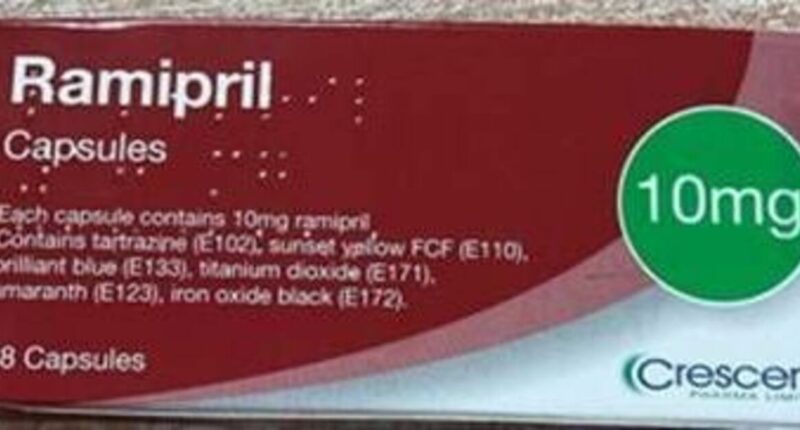
- The Medicines and Healthcare products Regulatory Agency has issued an urgent recall for Ramipril 10mg Capsules after a serious packaging error was discovered.
- Crescent Pharma Limited is recalling batch GR174091 after finding that some cartons labelled as containing 10mg capsules actually contained 5mg capsules instead. The MHRA said on Monday: “Crescent Pharma Limited has received one complaint to date, where it has been identified that, inside a sealed carton of Ramipril 10 mg Capsules Batch No.: GR174091, one blister pack of Ramipril 5 mg Capsules Batch No.: GR164094 was found.”
- Healthcare professionals have been instructed to immediately stop supplying the affected batch and quarantine all remaining stock. Pharmacies must contact patients who received the medication between 13 May 2025 and 16 April 2026.
- Patients prescribed Ramipril 10mg should check their medication packaging for batch number GR174091. The MHRA advised: “If the carton contains blister strips that are labelled and contain Ramipril 5 mg capsule, contact your dispensing pharmacy in the first instance.”
- Both medication strengths treat high blood pressure, heart failure and kidney disease, with the MHRA noting that any effects from reduced dosage would be gradual rather than life-threatening. Patients with concerns should contact their pharmacy or GP immediately.
- Anyone experiencing adverse reactions should report them through the MHRA Yellow Card scheme.
DISCOVER THE DETAILS: Important alert issued for Ramipril blood pressure medication, prompting patients to reach out to their pharmacy immediately.