Share this @internewscast.com
A health alert has been raised over a specific item on Tesco’s shelves, with stern warnings from officials instructing consumers not to eat the product.
Authorities from the Food Standards Agency (FSA) have sounded an alarm, compelling anyone who has this food item at home to return it immediately for safety reasons.
The caution concerns Le Superbe Raclette cheese, produced by Castelli UK Ltd, which may contain harmful Listeria monocytogenes bacteria. The warning is limited to Tesco stores, according to officials.
Product details
Le Superbe Raclette
| Pack size | 150g |
|---|---|
| Best before | 12 May 2025 |
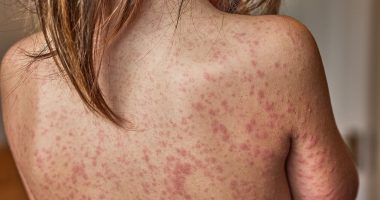
Listeria can trigger severe infections and in extreme instances, can be life-threatening. The FSA has commented: “Possible presence of Listeria monocytogenes in the above product. Symptoms caused by this organism can be similar to flu and include high temperature, muscle ache or pain, chills, feeling or being sick and diarrhoea. However, in rare cases, the infection can be more severe, causing serious complications, such as meningitis.”
The agency also highlighted that certain groups are at a higher risk of Listeria infection, including those over 65 years old, pregnant women and their unborn babies, newborns under one month, and individuals with compromised immune systems.
Tesco has displayed notices in affected stores advising customers to return the cheese product. The FSA added: “If you have bought this product do not eat it. Instead, return it to where it was purchased for a full refund. For more information contact 01732 835446.”
Symptoms of listeriosis
In most people, listeriosis has no symptoms or only causes mild symptoms for a few days, such as:
- a high temperature
- aches and pains
- chills
- feeling or being sick
- diarrhoea
Risks of listeriosis
Listeriosis is not usually serious for most people.
But some people have a higher risk of serious problems, including:
- anyone who is pregnant
- newborn babies
- people with a condition that weakens their immune system, such as cancer, diabetes, liver disease or kidney disease
- people having treatment that weakens their immune system, such as chemotherapy or steroid tablets
Older people are also at higher risk of getting seriously ill from listeriosis, and this risk increases with age. If you get listeriosis while you’re pregnant, there is a risk it could cause miscarriage or stillbirth.
In babies and people with a weakened immune system, listeriosis can sometimes lead to serious and life-threatening problems such as sepsis or meningitis.
According to UK Health Security Agency (UKHSA) data, rare listeriosis infections were almost a fifth higher than levels before the pandemic, with 179 reported cases in England and Wales in 2024, including 28 deaths.
The infection is contracted by eating listeria-contaminated food, and the health agency explained certain foods carry a higher risk, including soft cheeses, pate, smoked fish, chilled sliced meats and other chilled ready-to-eat products. Symptoms normally include a fever, aches and pains, nausea, vomiting, and diarrhoea, according to the NHS, but it can be life-threatening to the vulnerable.
About product recalls and withdrawals
If there is a problem with a food product that means it should not be sold, then it might be ‘withdrawn’ (taken off the shelves) or ‘recalled’ (when customers are asked to return the product). The FSA issues Product Recall Information Notices to let consumers and local authorities know about problems associated with food. In some cases, a ‘Food Alert for Action’ is issued. This provides local authorities with details of specific action to be taken on behalf of consumers. For more information click here.